Global clinical trials are scaling faster than ever in 2026, with sponsors pushing into new regions to accelerate drug development and meet patient needs. Yet the one element that consistently threatens timelines and approvals is the quality of Clinical Service Document Translation. A single imprecise term in an informed consent form or protocol amendment can trigger ethics committee rejections, force costly resubmissions, or even compromise participant safety.
The stakes are high because regulators now demand more than literal word-for-word conversions. They expect documents that read naturally in every target language while preserving exact scientific intent, legal protections, and cultural relevance.
Core linical Document Types That Require Professional Translation
Every trial generates dozens of materials that must cross borders without distortion. The most critical categories include:
Informed Consent Forms (ICFs) and pediatric assent documents
Full study protocols and all amendments
Investigator’s Brochures detailing the investigational product
Case Report Forms (CRFs), patient diaries, and electronic data capture templates
Patient-Reported Outcome (PRO) questionnaires and quality-of-life assessments
Adverse event reporting forms and safety update summaries
Recruitment advertisements, participant information sheets, and FAQs
Regulatory submission packages for FDA, EMA, or local authorities
Site training slides, SOPs, and monitoring guides
Product labels, dosing instructions, and patient leaflets
These aren’t optional add-ons. Regulators treat them as essential documents under ICH GCP E6 standards, and any language shortfall can stop site activation cold.
Translation Standards That Deliver Compliance in 2026
Reliable Clinical Service Document Translation follows a rigorous, auditable process rather than relying on speed or automation alone. Leading practices include:
Forward translation by medically qualified linguists, followed by independent back-translation and reconciliation
Linguistic validation and cognitive debriefing sessions with target-language speakers to confirm real-world understanding
Full traceability through Certificates of Translation (CoT) for every critical file
Strict terminology management across all documents to prevent drift in adverse-event language or dosing details
The FDA’s 21 CFR Part 50 guidance stresses that consent materials must be “understandable” to participants, while the EMA’s Clinical Trials Regulation requires participant-facing content in local languages with clear lay summaries. ISO 17100-certified workflows provide the quality backbone, ensuring every step—from project briefing to final sign-off—is documented and reviewable.
When these standards are met, trials move forward without the usual back-and-forth that eats months of valuable time.
What Happens When Accuracy Slips: Regulatory Realities and Hidden Costs
Industry data shows translation shortfalls are far from rare. Analyses indicate they factor into a substantial share of compliance setbacks, with some European pharmaceutical reviews linking over 60% of regulatory failures to unclear documentation language. Daily trial burn rates can range from $600,000 to $8 million once delays hit, according to Tufts Center research.
One older but telling example involved a Phase I study where a French-translated consent form softened risk descriptions. Volunteers experienced severe reactions that could have been anticipated with clearer language, leading to immediate suspension and lasting reputational damage.
More common today are quieter failures that never reach public headlines but still cost sponsors dearly.
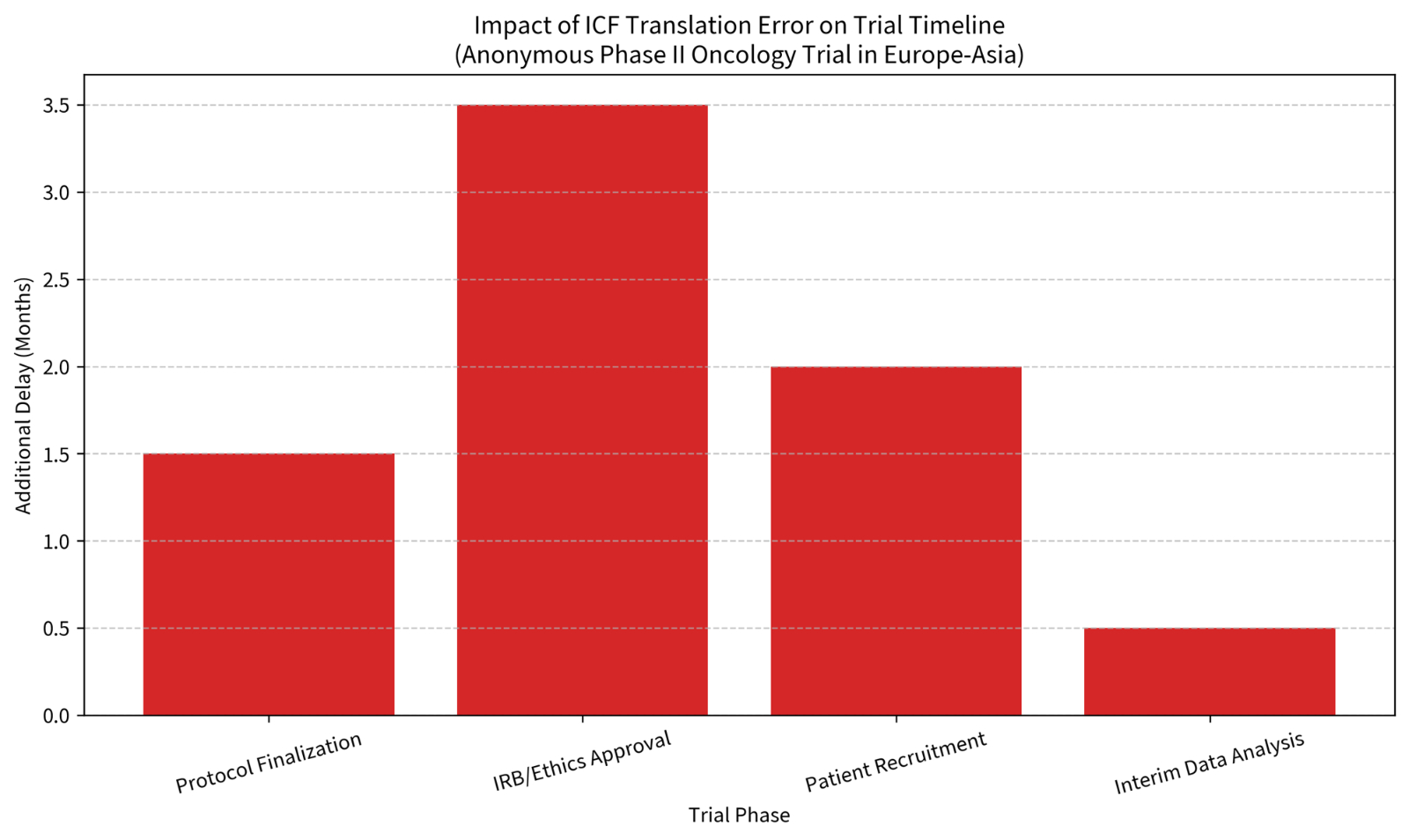
Anonymous Case Study 1 – Phase II Oncology Trial (Europe–Asia sites)A biotech sponsor submitted translated ICFs and protocol summaries for multi-country ethics review. Subtle phrasing differences around risk probabilities triggered repeated questions from two IRBs. The resulting revisions and re-approvals pushed site activation back by months and delayed the first-patient-in milestone.
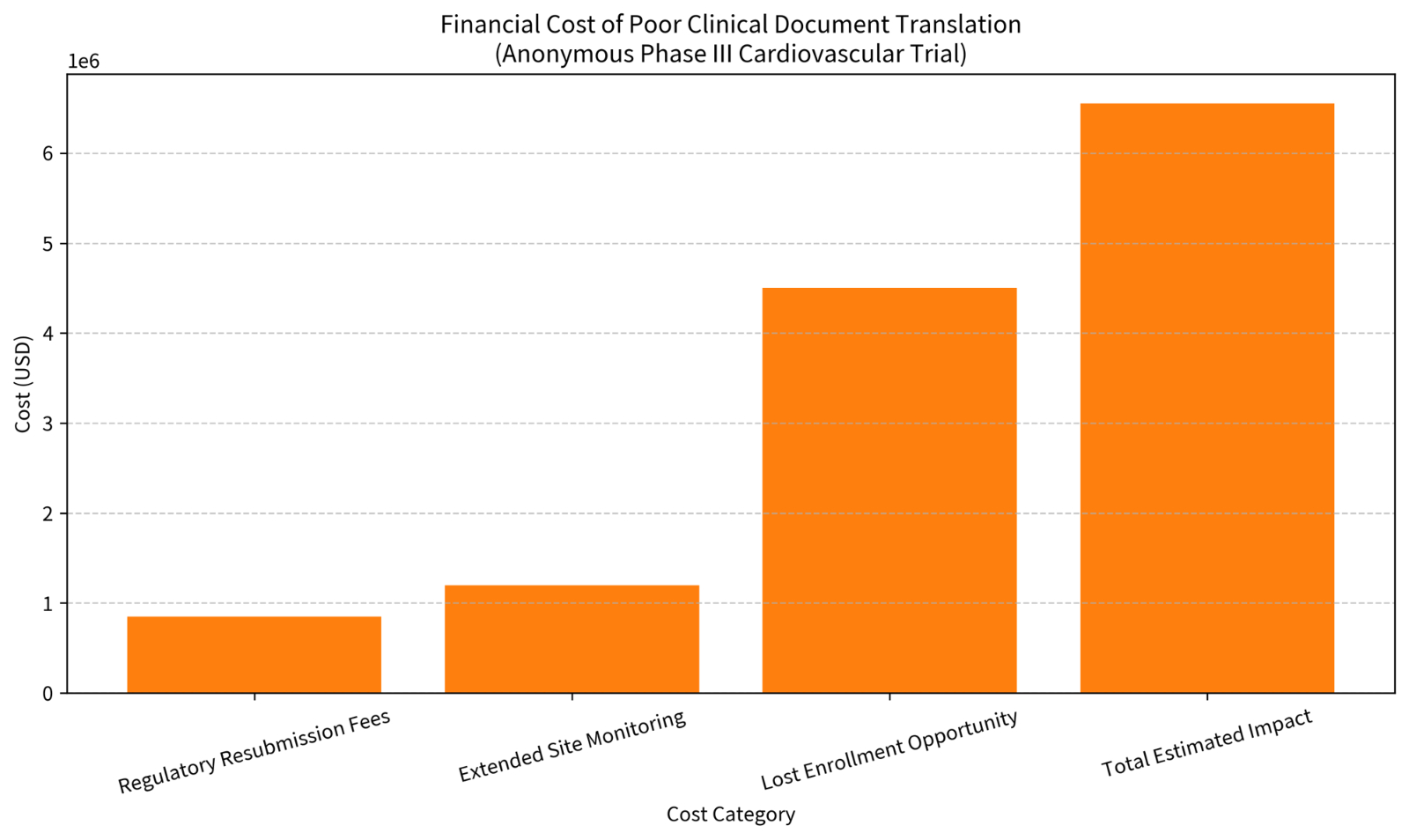
Anonymous Case Study 2 – Phase III Cardiovascular StudyProtocol amendments and PRO instruments contained inconsistent terminology across five languages. Ethics committees flagged discrepancies during review, forcing resubmissions, extra monitoring visits, and lost recruitment windows. The direct and indirect financial impact exceeded $6.5 million.
These aren’t hypothetical warnings. They reflect patterns that trial managers still encounter when choosing the wrong partner for Clinical Service Document Translation.
Clinical Document Translation – Which Provider Actually Delivers Accuracy?
The question every sponsor asks is simple: “Clinical document translation which one is accurate?” The answer lies in partners who treat medical content as a regulatory asset rather than a commodity. Look for teams with native medical linguists, documented validation processes, and a history of zero-defect submissions across FDA and EMA jurisdictions. Avoid providers who skip cognitive debriefing or rely heavily on unverified machine output for patient-facing materials.
When the right expertise is engaged early, revisions drop, approval cycles shorten, and trials stay on budget.
For organizations that need this level of precision without compromise, Artlangs Translation brings proven depth. Proficient across more than 230 languages and honed over years of specialized work, the team excels in clinical translation alongside video localization for training modules, short drama subtitle localization for patient education, game localization for interactive simulations, multilingual dubbing of short dramas and audiobooks on health topics, plus extensive multilingual data annotation and transcription. Their track record of successful global cases shows exactly how meticulous, compliant delivery keeps complex trials moving forward on schedule and fully regulator-ready.
That combination of breadth, depth, and real-world results makes all the difference when accuracy is non-negotiable.











