As healthcare systems worldwide prepare for expanded global trials and patient data sharing in 2026, Medical Report Translation & Proofreading has emerged as the non-negotiable layer that keeps critical clinical information accurate across borders. A single mistranslated lab value, dosage instruction, or diagnostic note in a patient discharge summary or pathology report can lead to delayed care, incorrect treatment, or worse. Hospitals, pharmaceutical sponsors, and contract research organizations know this risk all too well: one overlooked error in a translated medical report often triggers regulatory rework, extended review cycles, or even patient safety incidents that could have been avoided.
The stakes keep rising. The life sciences translation services market, already valued at USD 1.70 billion in 2025, is forecast to hit USD 1.84 billion this year and climb toward USD 3.27 billion by 2033, growing at a steady 8.55% CAGR. That expansion mirrors the surge in multinational clinical trials, cross-border electronic health records, and the push for inclusive patient recruitment. Yet the human cost of cutting corners remains painfully real. Studies consistently show that inaccurate medical interpretation or translation affects up to 30% of key communication units, with nearly 7% of those errors carrying moderate to high clinical significance. Professional processes slash that risk dramatically—ad-hoc or single-pass translations can double the odds of consequential mistakes.
The Hidden Danger of “Good Enough” Translation
Picture a routine oncology follow-up report translated from German to English for an international trial site. The source text describes “moderate response with residual disease at 2.3 cm.” A lone translator renders it as “partial improvement, tumor still around 2 cm.” Close enough? Not to the FDA reviewer who later flags inconsistency with the original imaging data. The sponsor scrambles to re-analyze scans, delays the data lock by weeks, and watches thousands in additional monitoring costs evaporate. Stories like this repeat across hospitals and sponsors every month. In one Pennsylvania analysis of patient safety events, language-related issues appeared in over 82% of reported incidents involving limited-English-proficiency patients, often resulting in missed or delayed care.
These aren’t abstract statistics. They translate into real-world consequences: prolonged hospital stays, medication errors, unnecessary procedures, and eroded trust between providers and patients. Regulatory bodies on both sides of the Atlantic demand linguistic precision in every submission—whether it’s an IND package, a post-market surveillance report, or a multilingual informed consent form. A single translation pass simply cannot guarantee the consistency, nuance, and cultural appropriateness that these documents require.
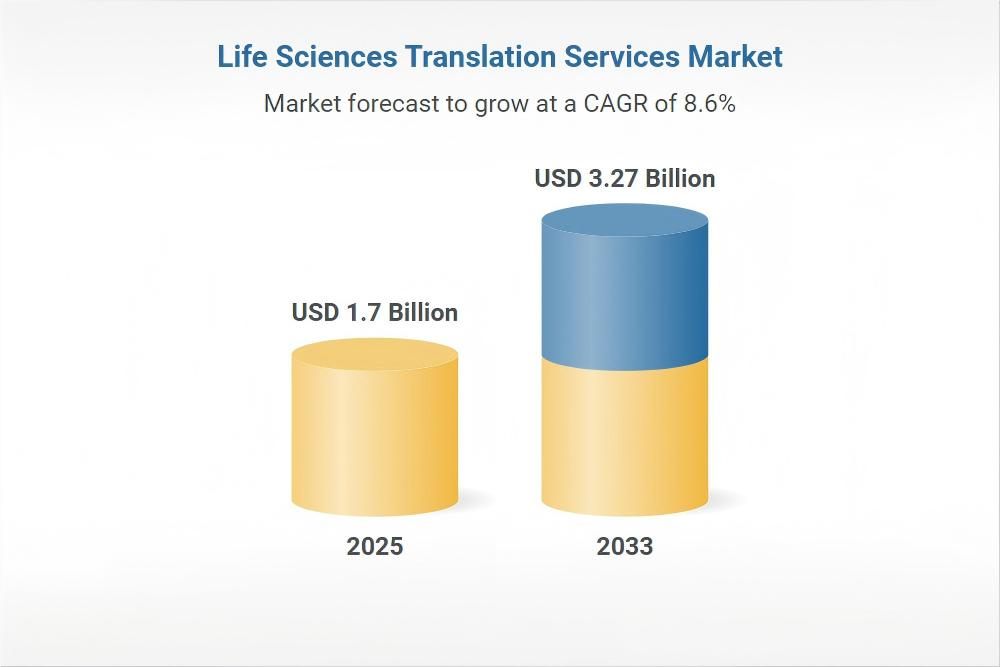
The chart above illustrates why 2026 is a pivotal year. Demand for accurate medical translation is accelerating in lockstep with global medicine use and trial complexity. Relying on machine tools or rushed freelancers without layered checks is no longer a viable shortcut.
The Gold Standard: A Proven Three-Review Process
Leading medical translation teams have long relied on a disciplined three-step workflow—commonly known as TEP (Translation, Editing, Proofreading)—to eliminate the gaps that single translators inevitably leave behind. Here’s how it works in practice, step by step:
Step 1 – Specialized TranslationA translator who is not only linguistically fluent but also holds domain credentials (often a medical degree, nursing background, or years of clinical translation experience) produces the first full draft. They work with approved terminology glossaries, reference the latest ICH guidelines, and flag any ambiguities back to the client for clarification. This isn’t word-for-word swapping; it’s meaning-for-meaning transfer that preserves clinical intent.
Step 2 – Independent EditingA second linguist, equally qualified and working blind to the original translator’s choices, reviews the entire document against the source. Editors focus on accuracy, completeness, and consistency. They catch subtle shifts in tone (critical in sensitive oncology or psychiatry reports), verify measurement units, drug names, and anatomical terminology, and ensure regulatory phrasing aligns with target-market requirements. This step typically reduces error rates by more than half.
Step 3 – Final ProofreadingThe third reviewer examines the document in isolation, focusing solely on the target language. They polish flow, check formatting (tables, footnotes, headings), confirm cultural appropriateness, and run a final consistency pass across the full set of reports for a single trial. Only after this triple layer does the file receive final sign-off and delivery.
To visualize how these steps interlock and catch issues at every stage, consider the standard three-review workflow used by top-tier providers:
(While the image above highlights accuracy gains in medical AI contexts, the same principle applies to human TEP workflows: layered checks compound reliability.)
This isn’t bureaucracy. It’s the difference between a report that regulators accept on first submission and one that triggers a complete response letter. Independent research confirms that professional multi-step processes cut clinically significant errors to roughly half the rate seen with ad-hoc or single-pass work.
Proofreading Standards That Actually Protect Patients
Beyond the three-review structure, elite providers follow additional quality gates tailored to medical content:
Terminology Management: Live, client-specific glossaries updated with every new modality—ADC payloads, gene-editing vectors, or companion diagnostics.
Back-Translation & Reconciliation (when required): Especially for informed consent or patient-facing materials.
Style & Formatting Validation: Ensuring the translated report mirrors the source layout so physicians can locate information instantly.
ISO 17100 & Client SOP Alignment: Full audit trails, version control, and documented corrective actions.
These standards aren’t marketing fluff. They are what regulators expect and what experienced sponsors now demand before sending documents to EMA, FDA, or PMDA reviewers.
Why the Extra Layer Pays for Itself Many Times Over
Every hour saved in regulatory review or avoided rework more than offsets the investment in proper Medical Report Translation & Proofreading. Sponsors who adopt rigorous TEP workflows report fewer queries, faster data locks, and smoother global site activation. Most importantly, they protect the people at the center of every trial: the patients.
When your next protocol amendment, safety update, or multinational patient report needs to cross language lines without losing a single decimal point or clinical nuance, the choice is clear. Precision at this level requires more than good intentions—it requires a partner who treats every medical document with the same rigor the original authors applied in the clinic or lab.
Artlangs Translation has built exactly that reputation over years of focused work in translation services, video localization, short drama subtitle localization, game localization, multi-language dubbing for short dramas and audiobooks, plus multi-language data annotation and transcription. Proficient across more than 230 languages and backed by hundreds of successful cases, their teams deliver the double-checked accuracy that turns complex medical reports into trusted assets—helping sponsors, hospitals, and research teams move forward with confidence in 2026 and beyond.











