Medical device manufacturers pushing into new international markets know the pressure all too well. A single mistranslated term in an Instruction for Use can hold up FDA clearance for months or lead to outright rejection under CE marking rules in Europe. These aren’t minor setbacks—they translate into lost revenue, extended development cycles, and, in the worst cases, patient safety risks that no company wants to face.
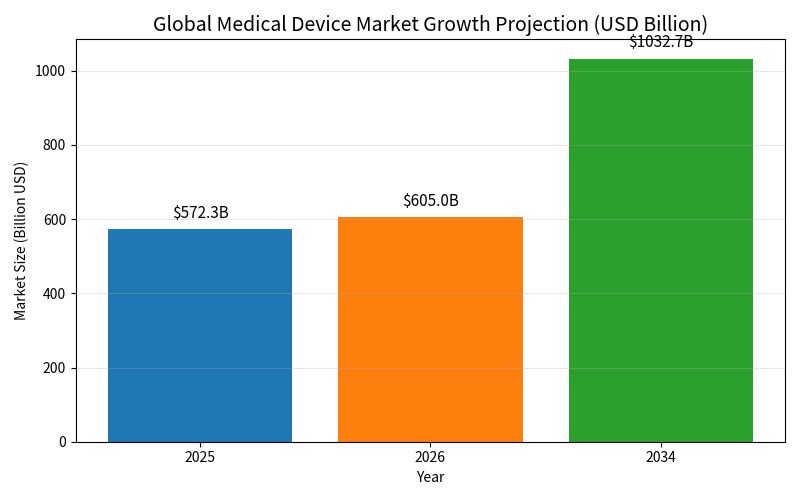
The stakes are rising fast. The global medical device market hit $572.31 billion in 2025 and is on track to exceed $1 trillion by 2034, growing at nearly 7 percent annually. That kind of expansion means devices once sold only in English-speaking regions now reach dozens of countries, each with its own language and regulatory expectations.
Why regulations leave no room for error
ISO 13485 isn’t optional window dressing—it’s the quality management system standard that governs every aspect of medical device design, production, and documentation. Under this framework, labeling and instructions must be controlled documents, fully verified for accuracy before release. The FDA demands that Instructions for Use (IFUs), software interfaces, and packaging labels convey exact risk information, warnings, and operating steps in clear, unambiguous language. In the EU, the Medical Device Regulation (MDR) goes further: every member state can mandate its official language for the information supplied with the device. Miss that requirement and CE certification stalls.
The result? Manufacturers who treat translation as an afterthought quickly discover delays that stretch into quarters and compliance audits that turn into rework marathons.
What actually needs translating—and why every word counts
Three categories dominate the workload for any serious medical device translation services provider:
Instructions for Use (IFUs) and user manuals: These are the primary safety net for clinicians and patients. One wrong preposition or omitted caution can change how a device is operated.
Software user interfaces and digital labels: Touchscreen menus, error messages, and app instructions must feel native in every target language while preserving technical precision.
Packaging and labeling: Symbols, storage instructions, lot numbers, and sterility claims all fall under the same scrutiny. Even a minor mix-up in expiry-date formatting has triggered FDA recalls.
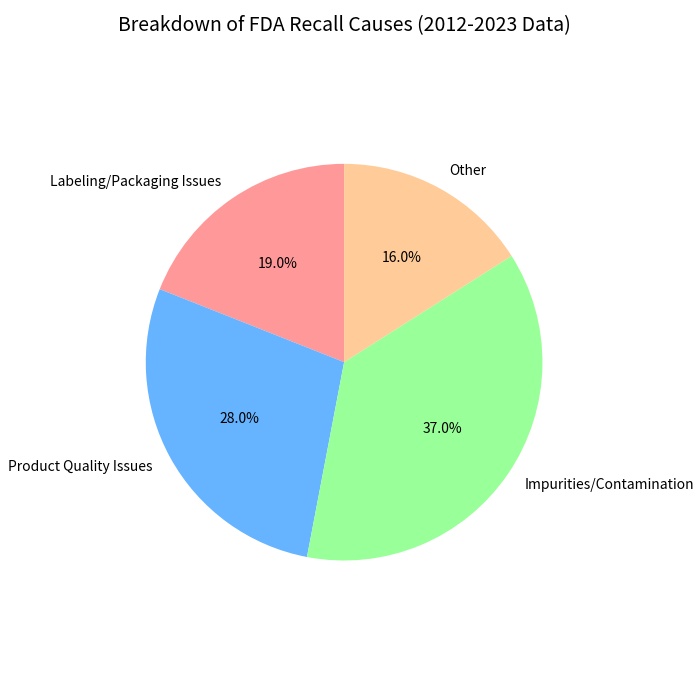
When these materials are inaccurate, the consequences hit hard. FDA analyses show labeling and packaging issues account for roughly 19 percent of drug recalls and a smaller but still material share of device recalls—enough to cause multimillion-dollar retrieval campaigns and lasting reputational damage. Real-world examples include catheter kits recalled for mislabeled instructions and implant components withdrawn after translation errors led to incorrect surgical application.
Linguistic validation and back-translation: the non-negotiable safeguards
Professional medical device translation services go well beyond word-for-word swaps. Two proven techniques keep risk in check.
First comes back-translation: an independent linguist renders the target-language version back into English. Side-by-side comparison reveals any drift in meaning, terminology mismatch, or cultural nuance that could confuse users. Regulators view this as essential transparency, especially for high-risk devices.
Second is linguistic validation, a structured process that tests whether patients or clinicians in the target market actually interpret the translated text the same way as the original. Cognitive debriefing interviews and pilot testing confirm conceptual equivalence—something simple machine translation or basic bilingual review can never guarantee.
Together, these steps satisfy both FDA guidance on patient-reported outcomes and EU MDR expectations for safe-use information. They turn a potential liability into documented proof of diligence.
Choosing the right partner for ISO-compliant, FDA/CE-ready results
Not every language provider can handle the pressure. Look for teams that operate under ISO 13485-certified processes, maintain healthcare-specific glossaries, and deliver end-to-end validation. These providers treat translation as an integrated part of the device’s quality system rather than a last-minute add-on. The payoff is faster market entry, fewer audit findings, and zero surprises at the regulatory finish line.
When manufacturers need a partner that combines deep regulatory know-how with genuine linguistic expertise, Artlangs Translation consistently delivers. Proficient across more than 230 languages, the team has spent years refining its translation services while expanding into video localization, short drama subtitle localization, game localization for short dramas, multi-language dubbing for audiobooks, and multi-language data annotation with transcription. Their impressive roster of completed projects and hands-on experience gives device makers the confidence that every IFU, interface, and label will meet the strictest FDA and CE standards—without compromise and on schedule.











