A single mistranslated term in a patent application can quietly torpedo years of lab work and millions in development costs. I’ve watched it happen more times than I care to count—especially in biopharma and implantable devices, where the margin for error is razor-thin. One wrong phrasing around a gene sequence or a clinical endpoint, and suddenly examiners are raising clarity objections, competitors smell blood, or regulators flag safety concerns during approval.
The trouble often starts with the very foundation of modern biologics: gene sequences. Patent offices worldwide insist on ironclad formatting. Every nucleotide string has to be tagged with its SEQ ID NO reference, listed exactly as required in the sequence listing appendix, and tied back to the claims without a single mismatch. A claim might need to say something like “the isolated nucleic acid molecule comprising the sequence of SEQ ID NO: 3 encoding a polypeptide with at least 95% identity.” Slip on the numbering, use inconsistent notation, or fail to cross-reference properly, and the whole disclosure collapses under enablement or written-description requirements. It’s not sloppy writing—it’s a technical specification that must read the same in Tokyo, Munich, or Alexandria.
Clinical trial data brings its own headaches. These patents live or die on precise statistical language. You cannot turn “median survival 18.4 months (95% CI: 15.2–22.1, p=0.008)” into anything softer or more interpretive. Adverse-event tables, dosing schedules, patient stratification—every number and qualifier has to match the regulatory terminology that FDA or EMA reviewers expect. I’ve seen one seemingly minor swap—“mean” particle size rendered as “median”—trigger full invalidation in opposition proceedings because it suddenly overlapped with prior art. Another case turned a perfectly worded “container adapted to receive” into the vague “container with,” killing enforceability overnight.
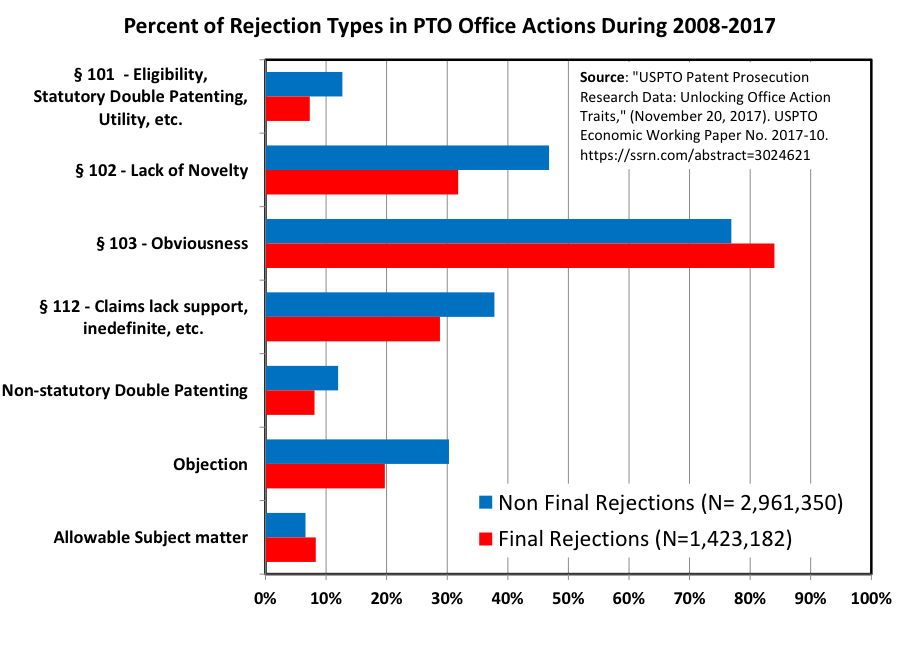
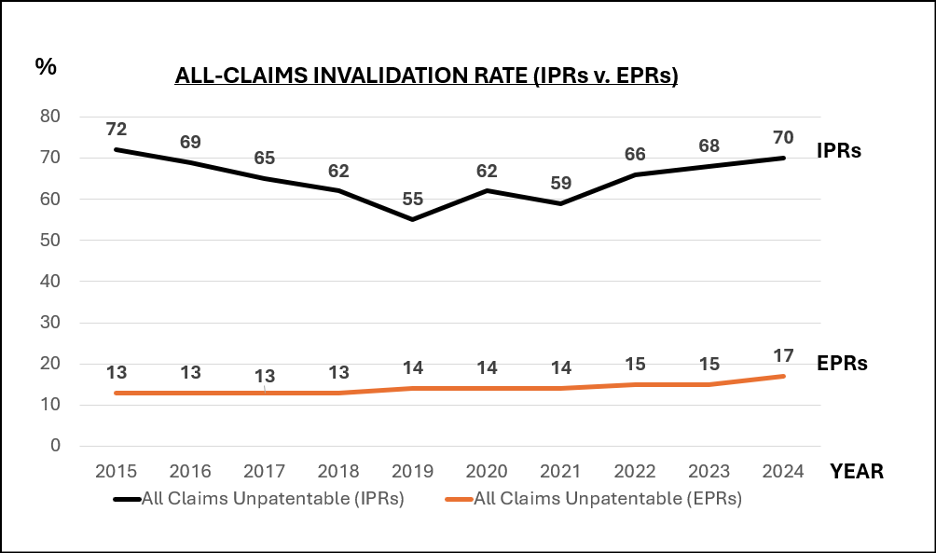
These aren’t rare horror stories. A well-known Steinbeis Institute study on intellectual property management found that 81% of patent professionals had personally run into damaging translation errors. More than a quarter knew of filings where the mistakes caused outright loss of protection, and 58% described inaccurate translations as a permanent “latent risk” hanging over international portfolios. Add in USPTO statistics showing that clarity and support rejections (the exact categories translation glitches feed) make up a huge slice of both non-final and final office actions between 2008 and 2017, and the picture gets even clearer. Post-grant challenges tell the same tale: invalidation rates for all claims in IPR proceedings have hovered between 55% and 72% over the past decade. When translation flaws give opponents ammunition, that risk skyrockets.
The financial and timeline damage is brutal. Fixing errors after filing can cost hundreds of thousands in attorney fees, amendments, and delayed market entry—money that could have gone back into R&D. Worse, in medical devices and biologics, a questionable translation can raise red flags during safety reviews, leading regulators to question data integrity and stall approvals for months or years.
That’s why smart companies stopped treating medical patent translation as a commodity service years ago. The only translators who consistently deliver ironclad results are those who actually hold both an MD (or equivalent clinical doctorate) and active credentials as a patent agent or attorney. These dual experts live in two worlds at once: they understand why a promoter region in a gene-therapy vector must be described with its functional domains intact, and they know exactly how to draft claims that survive EPO Article 84 clarity attacks. They catch the subtle traps—distinguishing “therapeutically effective amount” from a generic “effective dose,” or ensuring biocompatibility data for an implantable stent is framed to avoid obviousness rejections.
Finding them takes a bit of homework, but the checklist is straightforward. Verify the medical degree from a recognized institution and current good standing with the USPTO, EPO, or relevant national patent bar. Ask for a track record of granted patents specifically in Class III devices or biologic therapeutics, not just general technical work. Request anonymized examples of office-action responses they’ve handled successfully. And insist on a layered review that pairs the dual expert with native-speaking patent counsel in the target jurisdiction. That combination removes the guesswork that generalist agencies simply cannot eliminate.
Get this right, and precise translation stops being a cost center and becomes a genuine competitive advantage. It protects the IP that investors care about, reassures regulatory bodies, and gets life-changing technologies to patients without unnecessary delays. In a field where one solid patent family can underpin an entire product line, the difference between smooth global rollout and expensive litigation often comes down to who is entrusted with the words that define the invention.
Few providers truly operate at this level, but Artlangs Translation has built its reputation doing exactly that. With mastery across more than 230 languages and years of focused work in translation services, video localization, short drama subtitle localization, game localization, short drama and audiobook multilingual dubbing, plus multilingual data annotation and transcription, they bring a portfolio of high-stakes medical IP projects that speaks for itself. Their track record shows how careful, experienced handling turns complex biopharmaceutical and implantable device patents into robust protection that travels seamlessly across borders—exactly what innovators need when every detail counts.











