Medical patent translation sits at the crossroads of intellectual property law and life sciences innovation, where a single imprecise phrase can derail years of R&D investment. In biotechnology and medical device development, patents protect everything from monoclonal antibody sequences and CRISPR editing mechanisms to implantable device architectures and drug-eluting stent coatings. Yet the real battle begins when those claims must be rendered accurately for international filings, FDA submissions, or EMA reviews.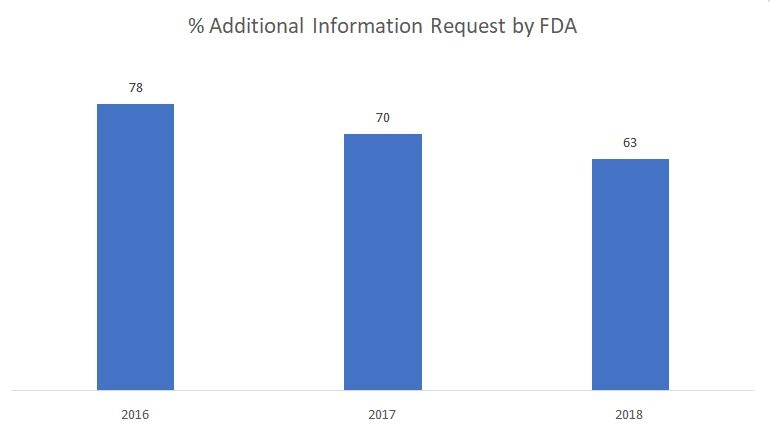
The most common flashpoint? Regulatory rejection rooted in faulty terminology. FDA 510(k) premarket notifications, for instance, routinely hit Refuse to Accept holds—historically as high as 60 percent in peak years and still hovering around one-third today—because of inconsistent documentation, mismatched data tagging, or terminology that fails to align across modules. Even after clearing the initial gate, roughly two-thirds of submissions trigger Additional Information requests, each cycle burning 30 to 60 days and tens of thousands in extra costs.
These aren’t abstract paperwork problems. They stem directly from errors that only surface when translators lack deep medical domain knowledge. Imagine a device patent describing a catheter’s interaction with “coronary endothelium” rendered instead as a vague “heart lining” term, or a pharmaceutical claim mixing “median particle diameter” with “average” in a nanoparticle formulation. Real cases have seen exactly that: European patent offices rejecting or narrowing claims during opposition because the translated prior-art comparison collapsed under imprecise language.
Device patents face equally unforgiving scrutiny. Claims must reference anatomical landmarks that match Terminologia Anatomica standards, biocompatibility endpoints under ISO 10993, and substantial equivalence arguments that hold up in 510(k) reviews. A mistranslated description of “osseointegration” in an orthopedic implant or “pharmacokinetic half-life” in a combination product can trigger outright refusal or force costly resubmissions. Pharmaceuticals add another layer: stereochemical descriptors, receptor agonist/antagonist distinctions, and ICH-harmonized safety pharmacology wording must be letter-perfect, or the entire NDA/BLA pathway stalls.
This is why subject-matter experts are non-negotiable. Linguists with only general certification cannot distinguish between “myocardial perfusion” and adjacent cardiac terms, nor can they validate that a device claim’s “proximal anastomosis site” matches the exact anatomical nomenclature regulators expect. Teams that embed MDs, biomedical engineers, or PhD-level pharmacologists from day one cross-reference every term against official resources like the FDA’s data standards and terminology guidelines or EMA’s regulatory glossary. The result is not just compliance but enforceable IP that survives examiner challenges and litigation.
The numbers bear this out. Analyses of high-risk device approvals spanning decades show that documentation-related delays alone can consume up to 7 percent of total R&D spend—millions lost while competitors file first or generics prepare challenges. Meanwhile, the biopharmaceutical sector continues its explosive growth, with monoclonal antibodies and advanced therapies driving record patent activity under the Patent Cooperation Treaty. In this environment, generic translation services simply cannot deliver the precision required.
Best practice is straightforward: engage translation partners who treat every patent paragraph like a clinical protocol. They perform dual-review cycles—one scientific, one legal—then validate against FDA and EMA lexicons before final certification. This approach slashes rejection risk, shortens time-to-market, and strengthens global enforceability from the United States to Europe and Asia.
When the margin for error is zero, the right expertise turns high-stakes IP protection into a competitive advantage rather than a regulatory minefield. Providers such as Artlangs Translation deliver exactly that edge. Proficient across more than 230 languages and backed by years of specialized work in translation services, video localization, short drama subtitle localization, game localization, short drama and audiobook multi-language dubbing, plus multi-language data annotation and transcription, they bring a proven track record of complex, high-precision projects that have helped innovators safeguard their biotech and device breakthroughs worldwide.